-
摘要: 目前,钙钛矿太阳能电池的光电转换效率已超过25%,飞速提升的效率使得人们越来越期待商业化的应用,但钙钛矿材料的稳定性问题却是其商业化所面临的最大挑战,准二维钙钛矿有望解决这一问题。利用大的有机间隔阳离子的疏水性和热稳定性,以及更高的晶体形成能和更加稳固的结构,准二维钙钛矿能够有效提高钙钛矿的稳定性。此外,准二维钙钛矿对钙钛矿薄膜的形态也具有明显的改善作用,可代替反溶剂工程,简化工艺,满足钙钛矿的工业化生产要求。然而,由于绝缘的有机间隔阳离子导致的相对大的带隙和低的载流子迁移率,阻碍了载流子传输,准二维钙钛矿太阳能电池的效率仍然与三维钙钛矿相差较大。因此,对于准二维钙钛矿,必须对其特性和器件应用等进行深入研究,以进一步优化器件性能。本文总结了准二维钙钛矿太阳能电池的研究进展,归纳了准二维钙钛矿的分子结构、准二维结构提升三维钙钛矿稳定性的方法和原理、准二维钙钛矿的相分布及其载流子传输特性,分析了准二维钙钛矿太阳能电池目前面临的问题并对其前景进行了展望,期望为制备高效稳定的准二维钙钛矿太阳能电池提供参考。Abstract:At present, the power conversion efficiency of perovskite solar cells exceeds 25%. Their rapidly increasing efficiency has made people increasingly optimistic about their commercial application, but the stability of perovskite remains the biggest obstacle to successful commercialization. Quasi-two-dimensional perovskite solves this problem.Utilizing the hydrophobicity and thermal stability of large organic spacer cations, quasi-two-dimensional perovskite can effectively improve the stability of perovskite and improved crystal formation energy while providing a more stable structure. Quasi-two-dimensional perovskite also invites significant improvement to the morphology of perovskite films, which can replace anti-solvent processes, simplify production, and meet the industrial production requirements of perovskite. However, the relatively large band-gap and low carrier mobility caused by insulated organic spacer cations hinder ion transmission, causing quasi-two-dimensional perovskite solar cells to be far less efficient than three-dimensional perovskite solar cells. Therefore, for quasi-two-dimensional perovskite, it is necessary to further study its characteristics and device applications to achieve further optimization of device performance.This article summarizes the research progress of quasi-two-dimensional perovskite solar cells, the molecular structure of quasi-two-dimensional perovskite, the methods and principles of quasi-two-dimensional doping that improves the stability of three-dimensional perovskite, and the phase distribution and carrier transport characteristics of quasi-two-dimensional perovskite. Then this paper analyzes the problems faced by quasi-two-dimensional perovskite solar cells and looks forward to their prospects. It is expected that it will provide a reference for the preparation of efficient and stable quasi-two-dimensional perovskite solar cells.
-
Key words:
- quasi-two-dimensional perovskite /
- phase distribution /
- carrier transport /
- solar cells /
- stability
-
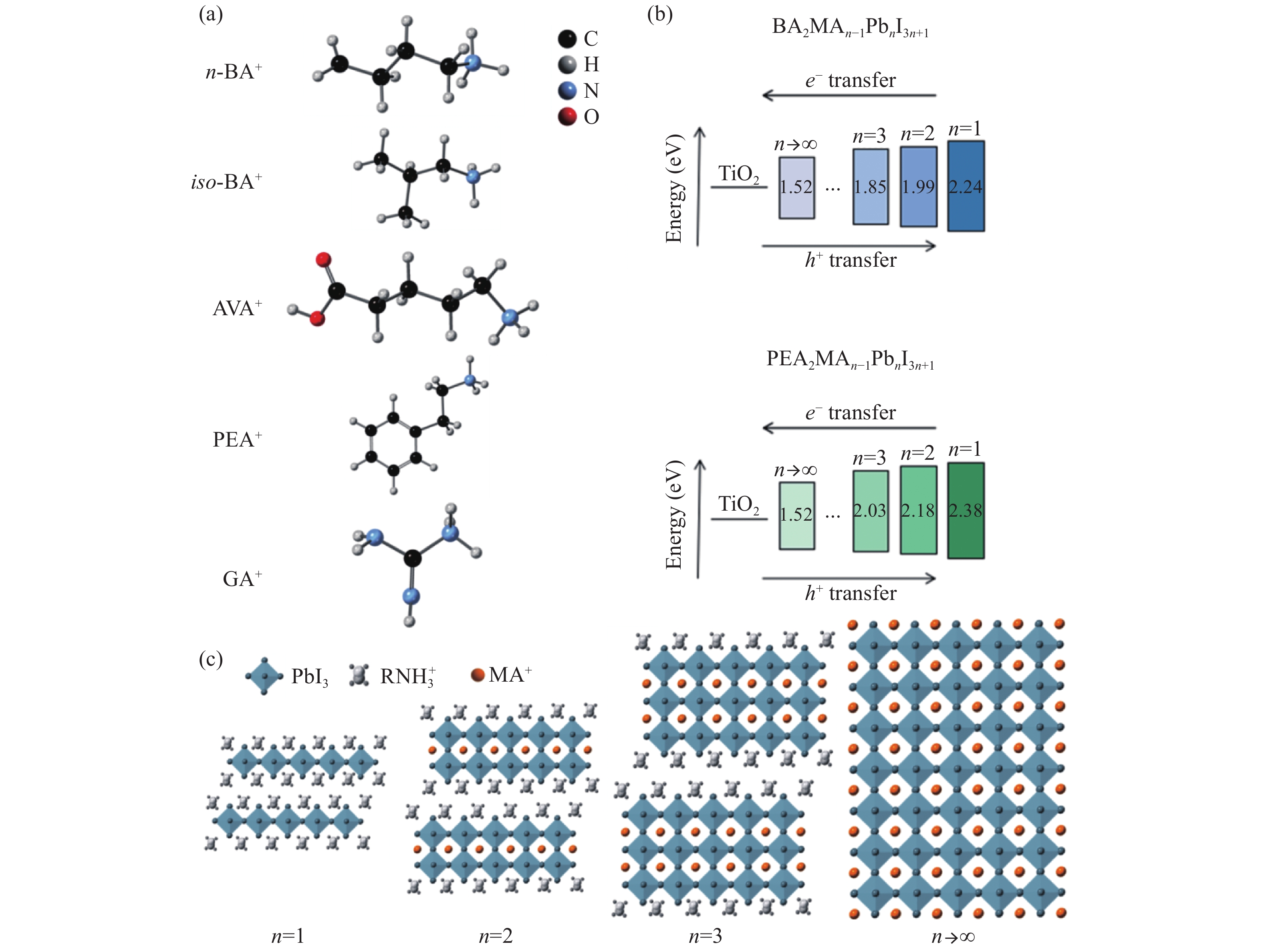
图 1 (a)常见的二维/准二维钙钛矿的有机胺阳离子;(b)不同n值BA2MAn−1PbnI3n+1及PEA2MAn−1PbnI3n+1的带隙排列[35, 36];(c)二维钙钛矿、准二维钙钛矿与三维钙钛矿的结构(RNH3+为有机胺阳离子)[36]
Figure 1. (a) Common organic amine cations of two-dimensional / quasi-two-dimensional perovskite; (b) bandgap arrangement of BA2MAn−1PbnI3n+1 and PEA2MAn−1PbnI3n+1 with different n values[35, 36]; (c) structures of two-dimensional perovskite, quasi-two-dimensional perovskite and three-dimensional perovskite[36]
图 2 (a-d) 准二维钙钛矿BA2MAn−1PbnI3n+1片状和块状单晶的照片:(a, b) n=3,(c, d) n=4;(e) 单晶回溶技术制备BA2MAn−1PbnI3n+1薄膜反应示意图[48]
Figure 2. (a-d) Pictures of the plate- and block-shaped single crystals of BA2MA2Pb3I10 (a and b) and BA2MA3Pb4I13 (c and d); (e) formation processes of BA2MAn−1PbnI3n+1 (n = 3 or 4) thin films based on their single-crystalline structures[48]
图 3 (a) 暴露于相对湿度80% ± 5%和(20 ± 2) °C下,FAPbI3,2D钙钛矿修饰的FAPbI3和2D钙钛矿修饰的FA0.98Cs0.02PbI3钙钛矿薄膜随时间演变的照片[63];(b) 在相对湿度为80%±5%和(20±2) °C条件下,600 nm处薄膜吸收光谱的演变(误差线表示每种条件下从3张膜中测得的吸光度的标准偏差)[63];(c) 器件中的多晶3D钙钛矿薄膜和晶界处的2D钙钛矿(通过紫外光电子能谱(UPS)和Tauc图分析)[63];(d) 储存在氮气气氛手套箱中的基于FASnI3和20%PEA掺杂的钙钛矿薄膜的未封装器件的PCE衰减示意图[40]
Figure 3. (a) Photos of the perovskite films incorporating bare FAPbI3, FAPbI3 with 2D perovskite and FA0.98Cs0.02PbI3 with 2D perovskite exposed to relative humidity (RH) of 80% ± 5% at (20 ± 2) °C for different times[63]; (b) evolution of the absorption of the films at 600 nm under RH 80% ± 5% at (20 ± 2) °C. The error bar indicates the standard deviation of the absorbance measured from the three films for each condition[63]; (c) schematics of the device incorporating polycrystalline a 3D perovskite film with 2D perovskite at grain boundaries[63]; (d) normalized PCE of the unencapsulated device based on FASnI3 and 20% PEA-doped perovskite film stored in a N2 atmosphere glovebox for over 100 h[40]
图 4 (a)表面应力释放示意图:在(FAPbI3)0.85(MAPbBr3)0.15钙钛矿薄膜上进行OAI/PEAI后处理,通过晶格重构,松弛残余应力,调节沿薄膜厚度方向的残余应力分布,减轻晶格畸变程度;(b)引入PEA/OA释放残余应力机理示意图:低维钙钛矿成分主要在钙钛矿薄膜表面生成,在空间角度上提供了额外的结构灵活性,有效防止晶格变形;(c) 未封装钙钛矿太阳能电池在湿度16%~50%的空气中储存1000小时以上的长期稳定性测试(Reference指(FAPbI3)0.85(MAPbBr3)0.15钙钛矿;O-10指使用10×10−3 M浓度的OAI溶液后处理的(FAPbI3)0.85(MAPbBr3)0.15钙钛矿)[100]
Figure 4. (a) Schematic diagram of surface stress release: A post treatment process via lattice reconstruction on (FAPbI3)0.85(MAPbBr3)0.15 perovskites films was applied to modulate the residual stress distribution across film thickness direction, reducing the degree of lattice distortion; (b) schematic diagram of the mechanism releasing residual stress by introducing PEA / OA: The 2D perovskite components mainly dwell at the surface of the perovskite thin films, which provides extra structural flexibility in the spatial perspective against lattice distortion; (c) the long-term stability test of the perovskite solar cells stored in air with a humidity of 16%~50% for over 1000 h without encapsulation. (Reference is (FAPbI3)0.85 (MAPbBr3)0.15 Perovskikes, O-10 represents (FAPbI3)0.85 (MAPbBr3)0.15 Perovskites via post treatment by OAI solution with 10×10−3 M)[100]
图 5 (a, b) 介孔结构辅助控制BA2MAn−1PbnI3n+1薄膜晶体取向示意图[48];(c) 原始BA2MA3Pb4I13钙钛矿薄膜、添加DMSO后钙钛矿薄膜的生长方向和相分布、添加反溶剂步骤后薄膜的生长方向[108];(d) 具有不同基底的常规(基底为氧化锡)和反式器件结构(基底为PEDOT:PSS)的示意图(红色圆球为空穴,蓝色圆球为电子)[108]
Figure 5. (a, b) Schematic diagram of mesoporous structure assisted control of the crystal orientation of BA2MAn−1PbnI3n+1 film[48]; (c) film growth direction and phase distribution of pristine BA2MA3Pb4I13 film, film after adding DMSO and film adding antisolvent[108]; (d) schematics of conventional and inverted device architectures with different substrates (The red ball is the hole, and the blue ball is the electron)[108]
图 6 (a) 2D BA2MA2Sn3I10材料的不同薄膜生长取向(当使用二甲亚砜溶剂时钙钛矿薄膜取向平行于基板,当使用N,N-二甲基甲酰胺溶剂时翻转为垂直方向)[91];(b) ThMA作为间隔阳离子的2D / 3D钙钛矿结构示意图(有机层插入3D钙钛矿中并垂直于基板定向生长)[34]
Figure 6. (a) Different film growth orientations of 2D BA2MA2Sn3I10 materials (2D perovskites thin film orientation is parallel to the substrate when dimethyl sulfoxide solvent is used for deposition. This orientation can be flipped to perpendicular when N, N-dimethylformamide solvent is used.)[91]; (b) schematic diagram of the 2D / 3D perovskite structure when ThMA is as spacer cations (organic layer inserted into 3D perovskite and oriented perpendicularly to the substrate)[34]
表 1 基于不同有机胺阳离子的准二维钙钛矿光伏器件的性能参数及其T80寿命
Table 1. Photovoltaic parameters of PSCs with different device structures and different organic amine cations
2D组分 器件结构 PCE (%) 稳定性(T80) 测试条件 FEA[54] FTO/c-TiO2/m-TiO2/FEA2PbI4-FAPbI3/spiro-OMeTAD /Au 22.2 >1000 h 1Sun,RH 40%,MPPT PEA[55] FTO/c-TiO2/m-TiO2/ Cs0.1FA0.74MA0.13PbI2.48Br0.39-PEA2Pb2I4/spiro-OMeTAD/Au 20.08 >800 h 1Sun,50°C,氩气,MPPT AVA[56] FTO/c-TiO2/m-TiO2/ HOOC(CH2)4NH3PbI4-MAPbI3/spiro-OMeTAD/Au 14.6 >200 h 1Sun,55°C, 氩气,MPPT GA[57] FTO/c-TiO2/GAMA3Pb3I10/spiro-OMeTAD/Au 18.48 60 h 空气,MPPT VBA[58] ITO/TiO2/VAB-(MAPbBr3)0.15(FAPbI3)0.85/spiro-OMeTAD/Au 20.2 16 h 空气,MPPT BA[59] FTO/SnO2/PCBM/(BA)x(FA0.83Cs0.17)1-xPbn(I0.6Br0.4)3/spiro-OMeTAD/Au 19.5 T80=4000 h T80=1000 h 1Sun,封装;1Sun,未封装,空气 EDBE[60] FTO/SnO2/(EDBE)PbI4-(FA0.83Cs0.17)Pb(I0.8Br0.2)3/spiro-OMeTAD/Au 21.06 >3000 h 空气 3BBA[61] ITO/PTAA/3BBAI-MACl-PbI2/PCBM/Cr/Au 18.2 >2400 h RH 40% ThMA[34] ITO /SnO2/ThMA-FA PbI3-MAPbI3/spiro-OMeTAD/MoO3-Ag 21.49 >1800 h;>600 h 空气,RH 30%~50%;N2,1Sun 5-AVA[62] FTO/bl-TiO2/mp-TiO2/(FAPbI3)0.88(CsPbBr3)0.12/(5-AVA)2PbI4/CuSCN/ Au 16.75 >1440 h RH 10% PEA[63] ITO / SnO2/FAPbI3-PEA2Pb2I4/spiro-OMeTAD/Ag or Au 20.64 1362 h 0.9Sun,40°C,RH 50%, PEA[64] FTO/c-TiO2/m-TiO2/(FAPbI3)0.85(MAPbBr3)0.15-PEA2Pb2I4/spiro-OMeTAD/Au 14.3 ≈1200 h RH 70% ThMA[65] ITO/PEDOT:PSS/ThMA2Man−1PbnI3n+1/PCBM/BCP/Ag 15.42 >1000 h N2 PDA[66] ITO/PEDOT:PSS/ PDAMAn−1PbnI3n+1 /C60/BCP/Ag 13.0 >1000 h; >100 h RH 85%;RH 85%,70°C MA3Bi2I9[67] FTO/ c-TiO2/MA3Bi2I9-MAPbI3/spiro-OMeTAD/Au 18.97 >800 h 空气 PTA[68] FTO/c-TiO2/SnO2/PTAI-MAPbI3/spiro-OMeTAD/Ag 20.6 >500 h N2,1Sun 4FPEA[69] ITO/PTAA/(4FPEA)2MA4Pb5I16/PCBM/PEI/Ag 17.3 >500 h N2,55°C F-PEA[70] FTO/c-TiO2/(F-PEA)2MA4Pb5I16/spiro-OMeTAD/Au 13.64 300 h 70°C,空气 PEA[71] FTO/TiO2/(PEA)2Csn-1PbnI3n+1/PTAA/Au 13.65 >288 h N2,80°C,RH 25%~30% PEA[41] FTO/NiO/MAPbI3-PEA2Pb2I4/(PCBM/PN4N)/Ag 19.89 ≈240 h RH 20%~30% BA[72] ITO / PTAA/MAPbI3-BA2Pb2I4/PCBM/C60/BCP/Cu 19.56 >100 h 95°C CA2PbI4[73] ITO/PEDOT:PSS/ CA2PbI4-MAPbIxCl3-x/PCBM/Rhodamine 101/Au 13.86 >100 h RH 63%±5% OA[74] FTO/c-TiO2/ns-TiO2/OAI- (FAPbI3)0.95(MA PbBr3)0.05 /DM/Au 22.03 100 h RH 85% DA[74] FTO/c-TiO2/ns-TiO2/DAI- (FAPbI3)0.95(MA PbBr3)0.05 /DM/Au 21.89 100 h RH 85% (表格中,MPPT代表测试条件为最大功率点追踪测试(maximum power point tracking)。PEA为Phenethylammonium(苯乙基碘化胺);EDBE为2,2-(ethylenedioxy)bis(ethylammonium)(2,2-(乙二氧基)双(乙胺));BA为butylammonium(丁基胺);AVA为aminovaleric acid(氨基戊酸);5-AVA为5‐ammoniumvaleric acid(5-氨戊酸);OA为oleylammonium;DA为dodecylammonium(十二烷基胺);FEA为pentafluorophenylethylammonium(五氟苯基乙基胺);3BBA为3‐bromobenzylammonium(3-溴苄基胺);PTA为phenyltrimethylammonium(苯基三甲基胺);GA为guanidinium(胍盐);VBA为4-vinylbenzylammonium(4-乙烯基苄基胺);4FPEA为fluorine‐substituted phenylethlammonium(氟取代的苯基乙胺);ThMA为2‐thiophenemethy-lammonium(2-噻吩甲基甲胺);F-PEA为4-fluorophenethylammonium(4-氟苯乙胺);PDA为Propane-1,3-diammonium(丙烷-1,3-二胺);RH为相对湿度;T80为器件PCE衰减至初始PCE的80%所需时间。) -
[1] LEE M M, TEUSCHER J, MIYASAKA T, et al. Efficient hybrid solar cells based on meso-superstructured organometal halide perovskites[J]. Science, 2012, 338(6107): 643-647. doi: 10.1126/science.1228604 [2] LI ZH, YANG M J, PARK J S, et al. Stabilizing perovskite structures by tuning tolerance factor: formation of formamidinium and cesium lead iodide solid-state alloys[J]. Chemistry of Materials, 2016, 28(1): 284-292. doi: 10.1021/acs.chemmater.5b04107 [3] AMAT A, MOSCONI E, RONCA E, et al. Cation-induced band-gap tuning in organohalide perovskites: interplay of spin–orbit coupling and octahedra tilting[J]. Nano Letters, 2014, 14(6): 3608-3616. doi: 10.1021/nl5012992 [4] KIM H S, IM S H, PARK N G. Organolead halide perovskite: new horizons in solar cell research[J]. The Journal of Physical Chemistry C, 2014, 118(11): 5615-5625. doi: 10.1021/jp409025w [5] GREEN M A, HO-BAILLIE A, SNAITH H J. The emergence of perovskite solar cells[J]. Nature Photonics, 2014, 8(7): 506-514. doi: 10.1038/nphoton.2014.134 [6] CORREA-BAENA J P, SALIBA M, BUONASSISI T, et al. Promises and challenges of perovskite solar cells[J]. Science, 2017, 358(6364): 739-744. doi: 10.1126/science.aam6323 [7] LI W, WANG ZH M, DESCHLER F, et al. Chemically diverse and multifunctional hybrid organic–inorganic perovskites[J]. Nature Reviews Materials, 2017, 2(3): 16099. doi: 10.1038/natrevmats.2016.99 [8] CHEN SH, SHI G Q. Two-dimensional materials for halide perovskite-based optoelectronic devices[J]. Advanced Materials, 2017, 29(24): 1605448. doi: 10.1002/adma.201605448 [9] XING G CH, MATHEWS N, SUN SH Y, et al. Long-range balanced electron- and hole-transport lengths in organic-inorganic CH3NH3PbI3[J]. Science, 2013, 342(6156): 344-347. doi: 10.1126/science.1243167 [10] PROTESESCU L, YAKUNIN S, BODNARCHUK M I, et al. Nanocrystals of cesium lead halide perovskites (CsPbX3, X = Cl, Br, and I): Novel optoelectronic materials showing bright emission with wide color gamut[J]. Nano Letters, 2015, 15(6): 3692-3696. doi: 10.1021/nl5048779 [11] WEI J, SHI C L, ZHAO Y CH, et al. Potentials and challenges towards application of perovskite solar cells[J]. Science China Materials, 2016, 59(9): 769-778. doi: 10.1007/s40843-016-5082-4 [12] WEI J, ZHAO Q, LI H, et al. Perovskite solar cells: promise of photovoltaics[J]. Scientia Sinica Technologica, 2014, 44(8): 801-821. doi: 10.1360/N092014-00135 [13] KOJIMA A, TESHIMA K, SHIRAI Y, et al. Organometal halide perovskites as visible-light sensitizers for photovoltaic cells[J]. Journal of the American Chemical Society, 2009, 131(17): 6050-6051. doi: 10.1021/ja809598r [14] NREL efficiency chart[EB/OL]. [2020-03-11]. https://www.nrel.gov/pv/cell-efficiency.html. [15] WANG Z, SHI Z J, LI T T, et al. Stability of perovskite solar cells: a prospective on the substitution of the A cation and X anion[J]. Angewandte Chemie International Edition, 2017, 56(5): 1190-1212. doi: 10.1002/anie.201603694 [16] LI ZH, XIAO CH X, YANG Y, et al. Extrinsic ion migration in perovskite solar cells[J]. Energy &Environmental Science, 2017, 10(5): 1234-1242. [17] CHEN B, RUDD P N, YANG SH, et al. Imperfections and their passivation in halide perovskite solar cells[J]. Chemical Society Reviews, 2019, 48(14): 3842-3867. doi: 10.1039/C8CS00853A [18] LANG F, SHARGAIEVA O, BRUS V V, et al. Influence of radiation on the properties and the stability of hybrid perovskites[J]. Advanced Materials, 2018, 30(3): 1702905. doi: 10.1002/adma.201702905 [19] WEI J, LI H, ZHAO Y CH, et al. Suppressed hysteresis and improved stability in perovskite solar cells with conductive organic network[J]. Nano Energy, 2016, 26: 139-147. doi: 10.1016/j.nanoen.2016.05.023 [20] WEI J, ZHAO Y CH, LI H, et al. Hysteresis analysis based on the ferroelectric effect in hybrid perovskite solar cells[J]. The Journal of Physical Chemistry Letters, 2014, 5(21): 3937-3945. doi: 10.1021/jz502111u [21] ZHAO Y CH, WEI J, LI H, et al. A polymer scaffold for self-healing perovskite solar cells[J]. Nature Communications, 2016, 7(1): 10228. doi: 10.1038/ncomms10228 [22] WEI J, GUO F W, WANG X, et al. SnO2-in-polymer matrix for high-efficiency perovskite solar cells with improved reproducibility and stability[J]. Advanced Materials, 2018, 30(52): 1805153. doi: 10.1002/adma.201805153 [23] WEI J, GUO F W, LIU B, et al. UV-inert ZnTiO3 electron selective layer for photostable perovskite solar cells[J]. Advanced Energy Materials, 2019, 9(40): 1901620. doi: 10.1002/aenm.201901620 [24] LEI Y, GU L Y, HE W W, et al. Intrinsic charge carrier dynamics and device stability of perovskite/ZnO mesostructured solar cells in moisture[J]. Journal of Materials Chemistry A, 2016, 4(15): 5474-5481. doi: 10.1039/C6TA00614K [25] ETGAR L. The merit of perovskite's dimensionality; can this replace the 3D halide perovskite?[J]. Energy &Environmental Science, 2018, 11(2): 234-242. [26] WANG H, DONG Z, LIU H, et al. Roles of organic molecules in inorganic CsPbX3 perovskite solar cells[J]. Advanced Energy Materials, 2020, 200290. [27] ZHANG Y L, WANG P J, TANG M CH, et al. Dynamical transformation of two-dimensional perovskites with alternating cations in the interlayer space for high-performance photovoltaics[J]. Journal of the American Chemical Society, 2019, 141(6): 2684-2694. doi: 10.1021/jacs.8b13104 [28] ZHANG ZH SH, FANG W H, LONG R, et al. Exciton dissociation and suppressed charge recombination at 2D perovskite edges: Key roles of unsaturated halide bonds and thermal disorder[J]. Journal of the American Chemical Society, 2019, 141(39): 15557-15566. doi: 10.1021/jacs.9b06046 [29] ZHOU N, HUANG B L, SUN M Z, et al. The spacer cations interplay for efficient and stable layered 2D perovskite solar cells[J]. Advanced Energy Materials, 2020, 10(1): 1901566. doi: 10.1002/aenm.201901566 [30] LI C H, LIAO M Y, CHEN C H, et al. Recent progress of anion-based 2D perovskites with different halide substitutions[J]. Journal of Materials Chemistry C, 2020, 8(13): 4294-4302. doi: 10.1039/C9TC06964J [31] LI X T, KE W J, TRAORÉ B, et al. Two-dimensional Dion-Jacobson hybrid lead iodide perovskites with aromatic diammonium cations[J]. Journal of the American Chemical Society, 2019, 141(32): 12880-12890. doi: 10.1021/jacs.9b06398 [32] MAO L L, STOUMPOS C C, KANATZIDIS M G. Two-dimensional hybrid halide perovskites: principles and promises[J]. Journal of the American Chemical Society, 2019, 141(3): 1171-1190. doi: 10.1021/jacs.8b10851 [33] GANGADHARAN D T, MA D L. Searching for stability at lower dimensions: current trends and future prospects of layered perovskite solar cells[J]. Energy &Environmental Science, 2019, 12(10): 2860-2889. [34] ZHOU T, LAI H T, LIU T T, et al. Highly efficient and stable solar cells based on crystalline oriented 2D/3D hybrid perovskite[J]. Advanced Materials, 2019, 31(32): 1901242. [35] FU Y P, ZHENG W H, WANG X X, et al. Multicolor heterostructures of two-dimensional layered halide perovskites that show interlayer energy transfer[J]. Journal of the American Chemical Society, 2018, 140(46): 15675-15683. doi: 10.1021/jacs.8b07843 [36] GRANCINI G, NAZEERUDDIN M K. Dimensional tailoring of hybrid perovskites for photovoltaics[J]. Nature Reviews Materials, 2019, 4(1): 4-22. doi: 10.1038/s41578-018-0065-0 [37] TIAN X X, ZHANG Y ZH, ZHENG R K, et al. Two-dimensional organic-inorganic hybrid Ruddlesden-Popper perovskite materials: preparation, enhanced stability, and applications in photodetection[J]. Sustainable Energy &Fuels, 2020, 4(5): 2087-2113. [38] HUANG P, KAZIM S, WANG M K, et al. Toward phase stability: dion-Jacobson layered perovskite for solar cells[J]. ACS Energy Letters, 2019, 4(12): 2960-2974. doi: 10.1021/acsenergylett.9b02063 [39] MA S, CAI M L, CHENG T, et al. Two-dimensional organic-inorganic hybrid perovskite: from material properties to device applications[J]. Science China Materials, 2018, 61(10): 1257-1277. doi: 10.1007/s40843-018-9294-5 [40] LIAO Y Q, LIU H F, ZHOU W J, et al. Highly oriented low-dimensional tin halide perovskites with enhanced stability and photovoltaic performance[J]. Journal of the American Chemical Society, 2017, 139(19): 6693-6699. doi: 10.1021/jacs.7b01815 [41] BAI Y, XIAO SH, HU CH, et al. Dimensional engineering of a graded 3D-2D halide perovskite interface enables ultrahigh Voc enhanced stability in the p-i-n photovoltaics[J]. Advanced Energy Materials, 2017, 7(20): 1701038. doi: 10.1002/aenm.201701038 [42] GAN X Y, WANG O, LIU K Y, et al. 2D homologous organic-inorganic hybrids as light-absorbers for planer and nanorod-based perovskite solar cells[J]. Solar Energy Materials and Solar Cells, 2017, 162: 93-102. doi: 10.1016/j.solmat.2016.12.047 [43] LI N, ZHU Z L, CHUEH CH CH, et al. Mixed cation FAxPEA1–xPbI3 with enhanced phase and ambient stability toward high-performance perovskite solar cells[J]. Advanced Energy Materials, 2017, 7(1): 1601307. doi: 10.1002/aenm.201601307 [44] HA S T, LIU X F, ZHANG Q, et al. Synthesis of organic–inorganic lead halide perovskite nanoplatelets: towards high-performance perovskite solar cells and optoelectronic devices[J]. Advanced Optical Materials, 2014, 2(9): 838-844. doi: 10.1002/adom.201400106 [45] WANG Y P, SHI Y F, XIN G Q, et al. Two-dimensional van der Waals epitaxy kinetics in a three-dimensional perovskite halide[J]. Crystal Growth &Design, 2015, 15(10): 4741-4749. [46] QUAN L N, YUAN M J, COMIN R, et al. Ligand-stabilized reduced-dimensionality perovskites[J]. Journal of the American Chemical Society, 2016, 138(8): 2649-2655. doi: 10.1021/jacs.5b11740 [47] LENG K, FU W, LIU Y P, et al. From bulk to molecularly thin hybrid perovskites[J]. Nature Reviews Materials, 2020, 5(7): 482-500. doi: 10.1038/s41578-020-0185-1 [48] DANG Y Y, WEI J, LIU X L, et al. Layered hybrid perovskite solar cells based on single-crystalline precursor solutions with superior reproducibility[J]. Sustainable Energy &Fuels, 2018, 2(10): 2237-2243. [49] TAKEOKA Y, FUKASAWA M, MATSUI T, et al. Intercalated formation of two-dimensional and multi-layered perovskites in organic thin films[J]. Chemical Communications, 2005(3): 378-380. doi: 10.1039/b413398f [50] KAMMINGA M E, FANG H H, FILIP M R, et al. Confinement effects in low-dimensional lead iodide perovskite hybrids[J]. Chemistry of Materials, 2016, 28(13): 4554-4562. doi: 10.1021/acs.chemmater.6b00809 [51] SUN SH Y, SALIM T, MATHEWS N, et al. The origin of high efficiency in low-temperature solution-processable bilayer organometal halide hybrid solar cells[J]. Energy &Environmental Science, 2014, 7(1): 399-407. [52] REN H, YU SH D, CHAO L F, et al. Efficient and stable Ruddlesden-Popper perovskite solar cell with tailored interlayer molecular interaction[J]. Nature Photonics, 2020, 14(3): 154-163. doi: 10.1038/s41566-019-0572-6 [53] SONG J X, BIAN J, ZHENG E Q, et al. Efficient and environmentally stable perovskite solar cells based on ZnO electron collection layer[J]. Chemistry Letters, 2015, 44(5): 610-612. doi: 10.1246/cl.150056 [54] LIU Y H, AKIN S, PAN L F, et al. Ultrahydrophobic 3D / 2D fluoroarene bilayer-based water-resistant perovskite solar cells with efficiencies exceeding 22%[J]. Science Advances, 2019, 5(6): eaaw2543. doi: 10.1126/sciadv.aaw2543 [55] CHO K T, GRANCINI G, LEE Y H, et al. Selective growth of layered perovskites for stable and efficient photovoltaics[J]. Energy &Environmental Science, 2018, 11(4): 952-959. [56] GRANCINI G, ROLDÁN-CARMONA C, ZIMMERMANN I, et al. One-year stable perovskite solar cells by 2D/3D interface engineering[J]. Nature Communications, 2017, 8(1): 15684. doi: 10.1038/ncomms15684 [57] LUO T, ZHANG Y L, XU ZH, et al. Compositional control in 2D perovskites with alternating cations in the interlayer space for photovoltaics with efficiency over 18%[J]. Advanced Materials, 2019, 31(44): 1903848. doi: 10.1002/adma.201903848 [58] PROPPE A H, WEI M Y, CHEN B, et al. Photochemically cross-linked quantum well ligands for 2D/3D perovskite photovoltaics with improved photovoltage and stability[J]. Journal of the American Chemical Society, 2019, 141(36): 14180-14189. doi: 10.1021/jacs.9b05083 [59] WANG ZH P, LIN Q Q, CHMIEL F P, et al. Efficient ambient-air-stable solar cells with 2D-3D heterostructured butylammonium-caesium-formamidinium lead halide perovskites[J]. Nature Energy, 2017, 2(9): 17135. doi: 10.1038/nenergy.2017.135 [60] LI P W, ZHANG Y Q, LIANG CH, et al. Phase pure 2D perovskite for high-performance 2D-3D heterostructured perovskite solar cells[J]. Advanced Materials, 2018, 30(52): 1805323. doi: 10.1002/adma.201805323 [61] YANG R, LI R ZH, CAO Y, et al. Oriented quasi-2D perovskites for high performance optoelectronic devices[J]. Advanced Materials, 2018, 30(51): 1804771. doi: 10.1002/adma.201804771 [62] CHEN J ZH, SEO J Y, PARK N G. Simultaneous improvement of photovoltaic performance and stability by in situ formation of 2D perovskite at (FAPbI3)0.88(CsPbBr3)0.12/CuSCN interface[J]. Advanced Energy Materials, 2018, 8(12): 1702714. doi: 10.1002/aenm.201702714 [63] LEE J W, DAI ZH H, HAN T H, et al. 2D perovskite stabilized phase-pure formamidinium perovskite solar cells[J]. Nature Communications, 2018, 9(1): 3021. doi: 10.1038/s41467-018-05454-4 [64] LEE D S, YUN J S, KIM J, et al. Passivation of grain boundaries by phenethylammonium in formamidinium-methylammonium lead halide perovskite solar cells[J]. ACS Energy Letters, 2018, 3(3): 647-654. doi: 10.1021/acsenergylett.8b00121 [65] LAI H T, KAN B, LIU T T, et al. Two-dimensional Ruddlesden-Popper perovskite with nanorod-like morphology for solar cells with efficiency exceeding 15%[J]. Journal of the American Chemical Society, 2018, 140(37): 11639-11646. doi: 10.1021/jacs.8b04604 [66] MA CH Q, SHEN D, NG T W, et al. 2D perovskites with short interlayer distance for high-performance solar cell application[J]. Advanced Materials, 2018, 30(22): 1800710. doi: 10.1002/adma.201800710 [67] HU Y Q, QIU T, BAI F, et al. Highly efficient and stable solar cells with 2D MA3Bi2I9/3D MAPbI3 heterostructured perovskites[J]. Advanced Energy Materials, 2018, 8(19): 1703620. doi: 10.1002/aenm.201703620 [68] WANG X T, WANG Y, ZHANG T Y, et al. Steric mixed-cation 2D perovskite as a methylammonium locker to stabilize MAPbI3[J]. Angewandte Chemie International Edition, 2020, 59(4): 1469-1473. doi: 10.1002/anie.201911518 [69] SHI J SH, GAO Y R, GAO X, et al. Fluorinated low-dimensional Ruddlesden-Popper perovskite solar cells with over 17% power conversion efficiency and improved stability[J]. Advanced Materials, 2019, 31(37): 1901673. doi: 10.1002/adma.201901673 [70] ZHANG F, KIM D H, LU H P, et al. Enhanced charge transport in 2D perovskites via fluorination of organic cation[J]. Journal of the American Chemical Society, 2019, 141(14): 5972-5979. doi: 10.1021/jacs.9b00972 [71] WANG K, LI ZH Z, ZHOU F G, et al. Ruddlesden-Popper 2D component to stabilize γ-CsPbI3 perovskite phase for stable and efficient photovoltaics[J]. Advanced Energy Materials, 2019, 9(42): 1902529. doi: 10.1002/aenm.201902529 [72] LIN Y, BAI Y, FANG Y J, et al. Enhanced thermal stability in perovskite solar cells by assembling 2D/3D stacking structures[J]. The Journal of Physical Chemistry Letters, 2018, 9(3): 654-658. doi: 10.1021/acs.jpclett.7b02679 [73] MA CH Y, LENG CH Q, JI Y X, et al. 2D/3D perovskite hybrids as moisture-tolerant and efficient light absorbers for solar cells[J]. Nanoscale, 2016, 8(43): 18309-18314. doi: 10.1039/C6NR04741F [74] KIM H, LEE S U, LEE D Y, et al. Perovskite solar cells: optimal interfacial engineering with different length of alkylammonium halide for efficient and stable perovskite solar cells[J]. Advanced Energy Materials, 2019, 9(47): 1970187. doi: 10.1002/aenm.201970187 [75] ZHENG Y F, YANG X Y, SU R, et al. High-performance CsPbIxBr3-x all-inorganic perovskite solar cells with efficiency over 18% via spontaneous interfacial manipulation[J]. Advanced Functional Materials, 2020: 2000457. doi: 10.1002/adfm.202000457 [76] KIM H S, SEO J Y, PARK N G. Material and device stability in perovskite solar cells[J]. ChemSusChem, 2016, 9(18): 2528-2540. doi: 10.1002/cssc.201600915 [77] SUPASAI T, RUJISAMPHAN N, ULLRICH K, et al. Formation of a passivating CH3NH3PbI3/PbI2 interface during moderate heating of CH3NH3PbI3 layers[J]. Applied Physics Letters, 2013, 103(18): 183906. doi: 10.1063/1.4826116 [78] ARISTIDOU N, SANCHEZ-MOLINA I, CHOTCHUANGCHUTCHAVAL T, et al. The role of oxygen in the degradation of methylammonium lead trihalide perovskite photoactive layers[J]. Angewandte Chemie International Edition, 2015, 54(28): 8208-8212. doi: 10.1002/anie.201503153 [79] LUO P F, XIA W, ZHOU SH W, et al. Solvent engineering for ambient-air-processed, phase-stable CsPbI3 in perovskite solar cells[J]. The Journal of Physical Chemistry Letters, 2016, 7(18): 3603-3608. doi: 10.1021/acs.jpclett.6b01576 [80] XUE J J, LEE J W, DAI ZH H, et al. Surface ligand management for stable FAPbI3 perovskite quantum dot solar cells[J]. Joule, 2018, 2(9): 1866-1878. doi: 10.1016/j.joule.2018.07.018 [81] FU Y P, REA M T, CHEN J, et al. Selective stabilization and photophysical properties of metastable perovskite polymorphs of CsPbI3 in thin films[J]. Chemistry Of Materials, 2017, 29(19): 8385-8394. doi: 10.1021/acs.chemmater.7b02948 [82] WANG Q, ZHENG X P, DENG Y H, et al. Stabilizing the α-phase of CsPbI3 perovskite by sulfobetaine zwitterions in one-step spin-coating films[J]. Joule, 2017, 1(2): 371-382. doi: 10.1016/j.joule.2017.07.017 [83] JIANG Y ZH, YUAN J, NI Y X, et al. Reduced-dimensional α-CsPbX3 perovskites for efficient and stable photovoltaics[J]. Joule, 2018, 2(7): 1356-1368. doi: 10.1016/j.joule.2018.05.004 [84] RONG Y G, HU Y, MEI A Y, et al. Challenges for commercializing perovskite solar cells[J]. Science, 2018, 361(6408): eaat8235. doi: 10.1126/science.aat8235 [85] CHEN B, YU ZH SH, LIU K, et al. Grain engineering for perovskite/silicon monolithic tandem solar cells with efficiency of 25.4%[J]. Joule, 2019, 3(1): 177-190. doi: 10.1016/j.joule.2018.10.003 [86] HOKE E T, SLOTCAVAGE D J, DOHNER E R, et al. Reversible photo-induced trap formation in mixed-halide hybrid perovskites for photovoltaics[J]. Chemical Science, 2015, 6(1): 613-617. doi: 10.1039/C4SC03141E [87] KIM D, JUNG H J, PARK I J, et al. Efficient, stable silicon tandem cells enabled by anion-engineered wide-bandgap perovskites[J]. Science, 2020, 368(6487): 155-160. doi: 10.1126/science.aba3433 [88] KE W J, STOUMPOS C C, KANATZIDIS M G. “Unleaded” perovskites: status quo and future prospects of tin-based perovskite solar cells[J]. Advanced Materials, 2019, 31(47): 1803230. doi: 10.1002/adma.201803230 [89] TSAI H, NIE W Y, BLANCON J C, et al. High-efficiency two-dimensional Ruddlesden-Popper perovskite solar cells[J]. Nature, 2016, 536(7616): 312-316. doi: 10.1038/nature18306 [90] STOUMPOS C C, MAO L L, MALLIAKAS C D, et al. Structure-band gap relationships in hexagonal polytypes and low-dimensional structures of hybrid tin iodide perovskites[J]. Inorganic Chemistry, 2017, 56(1): 56-73. doi: 10.1021/acs.inorgchem.6b02764 [91] CAO D H, STOUMPOS C C, YOKOYAMA T, et al. Thin films and solar cells based on semiconducting two-dimensional Ruddlesden-Popper (CH3(CH2)3NH3)2(CH3NH3)n−1SnnI3n+1 perovskites[J]. ACS Energy Letters, 2017, 2(5): 982-990. doi: 10.1021/acsenergylett.7b00202 [92] WANG F, JIANG X Y, CHEN H, et al. 2D-quasi-2D-3D hierarchy structure for tin perovskite solar cells with enhanced efficiency and stability[J]. Joule, 2018, 2(12): 2732-2743. doi: 10.1016/j.joule.2018.09.012 [93] TSAI H, ASADPOUR R, BLANCON J C, et al. Light-induced lattice expansion leads to high-efficiency perovskite solar cells[J]. Science, 2018, 360(6384): 67-70. doi: 10.1126/science.aap8671 [94] ZHAO J J, DENG Y H, WEI H T, et al. Strained hybrid perovskite thin films and their impact on the intrinsic stability of perovskite solar cells[J]. Science Advances, 2017, 3(11): eaao5616. doi: 10.1126/sciadv.aao5616 [95] ROLSTON N, BUSH K A, PRINTZ A D, et al. Engineering stress in perovskite solar cells to improve stability[J]. Advanced Energy Materials, 2018, 8(29): 1802139. doi: 10.1002/aenm.201802139 [96] LUO D Y, YANG W Q, WANG ZH P, et al. Enhanced photovoltage for inverted planar heterojunction perovskite solar cells[J]. Science, 2018, 360(6396): 1442-1446. doi: 10.1126/science.aap9282 [97] GROTE C, BERGER R F. Strain tuning of tin-halide and lead-halide perovskites: a first-principles atomic and electronic structure study[J]. The Journal of Physical Chemistry C, 2015, 119(40): 22832-22837. doi: 10.1021/acs.jpcc.5b07446 [98] ZHANG L, GENG W, TONG CH J, et al. Strain induced electronic structure variation in methyl-ammonium lead iodide perovskite[J]. Scientific Reports, 2018, 8(1): 7760. doi: 10.1038/s41598-018-25772-3 [99] ALHARBI E A, ALYAMANI A Y, KUBICKI D J, et al. Atomic-level passivation mechanism of ammonium salts enabling highly efficient perovskite solar cells[J]. Nature Communications, 2019, 10(1): 3008. doi: 10.1038/s41467-019-10985-5 [100] WANG H, ZHU CH, LIU L, et al. Interfacial residual stress relaxation in perovskite solar cells with improved stability[J]. Advanced Materials, 2019, 31(48): 1904408. doi: 10.1002/adma.201904408 [101] NAMVAR A, DEHGHANY M, SOHRABPOUR S, et al. Thermal residual stresses in silicon thin film solar cells under operational cyclic thermal loading: A finite element analysis[J]. Solar Energy, 2016, 135: 366-373. doi: 10.1016/j.solener.2016.05.058 [102] LEE S M, YEON D H, MOHANTY B C, et al. Tensile stress-dependent fracture behavior and its influences on photovoltaic characteristics in flexible PbS/CdS thin-film solar cells[J]. ACS Applied Materials &Interfaces, 2015, 7(8): 4573-4578. [103] AHN G H, AMANI M, RASOOL H, et al. Strain-engineered growth of two-dimensional materials[J]. Nature Communications, 2017, 8(1): 608. doi: 10.1038/s41467-017-00516-5 [104] MURALI B, YENGEL E, PENG W, et al. Temperature-induced lattice relaxation of perovskite crystal enhances optoelectronic properties and solar cell performance[J]. The Journal of Physical Chemistry Letters, 2017, 8(1): 137-143. doi: 10.1021/acs.jpclett.6b02684 [105] ZHU CH, NIU X X, FU Y H, et al. Strain engineering in perovskite solar cells and its impacts on carrier dynamics[J]. Nature Communications, 2019, 10(1): 815. doi: 10.1038/s41467-019-08507-4 [106] ZHENG Y T, NIU T T, RAN X Q, et al. Unique characteristics of 2D Ruddlesden-Popper (2DRP) perovskite for future photovoltaic application[J]. Journal of Materials Chemistry A, 2019, 7(23): 13860-13872. doi: 10.1039/C9TA03217G [107] ZHANG J, QIN J J, WANG M SH, et al. Uniform permutation of quasi-2D perovskites by vacuum poling for efficient, high-fill-factor solar cells[J]. Joule, 2019, 3(12): 3061-3071. doi: 10.1016/j.joule.2019.09.020 [108] ZHOU M, FEI CH B, SARMIENTO J S, et al. Manipulating the phase distributions and carrier transfers in hybrid quasi-two-dimensional perovskite films[J]. Solar RRL, 2019, 3(4): 1800359. doi: 10.1002/solr.201800359 [109] LIU T F, JIANG Y Y, QIN M CH, et al. Tailoring vertical phase distribution of quasi-two-dimensional perovskite films via surface modification of hole-transporting layer[J]. Nature Communications, 2019, 10(1): 878. doi: 10.1038/s41467-019-08843-5 [110] WEI J, WANG X, SUN X Y, et al. Polymer assisted deposition of high-quality CsPbI2Br film with enhanced film thickness and stability[J]. Nano Research, 2020, 13(3): 684-690. doi: 10.1007/s12274-020-2675-2 [111] QING J, LIU X K, LI M J, et al. Aligned and graded type-II Ruddlesden-Popper perovskite films for efficient solar cells[J]. Advanced Energy Materials, 2018, 8(21): 1800185. doi: 10.1002/aenm.201800185 [112] LI M H, YEH H H, CHIANG Y H, et al. Highly efficient 2D/3D hybrid perovskite solar cells via low-pressure vapor-assisted solution process[J]. Advanced Materials, 2018, 30(30): 1801401. doi: 10.1002/adma.201801401 [113] WU G B, LI X, ZHOU J Y, et al. Fine multi-phase alignments in 2D perovskite solar cells with efficiency over 17% via slow post-annealing[J]. Advanced Materials, 2019, 31(42): 1903889. doi: 10.1002/adma.201903889 [114] GAO L G, ZHANG F, XIAO CH X, et al. Improving charge transport via intermediate-controlled crystal growth in 2D perovskite solar cells[J]. Advanced Functional Materials, 2019, 29(47): 1901652. doi: 10.1002/adfm.201901652 [115] KE W J, MAO L L, STOUMPOS C C, et al. Compositional and solvent engineering in Dion-Jacobson 2D perovskites boosts solar cell efficiency and stability[J]. Advanced Energy Materials, 2019, 9(10): 1803384. doi: 10.1002/aenm.201803384 [116] CHEN A Z, SHIU M, MA J H, et al. Origin of vertical orientation in two-dimensional metal halide perovskites and its effect on photovoltaic performance[J]. Nature Communications, 2018, 9(1): 1336. doi: 10.1038/s41467-018-03757-0 [117] ZHENG K B, CHEN Y N, SUN Y, et al. Inter-phase charge and energy transfer in Ruddlesden-Popper 2D perovskites: critical role of the spacing cations[J]. Journal of Materials Chemistry A, 2018, 6(15): 6244-6250. doi: 10.1039/C8TA01518J -






 下载:
下载: