Synthesis of chitosan-based polymer carbon dots fluorescent materials and application of self-assembled drug-loading
doi: 10.3788/CO.20181103.0420
-
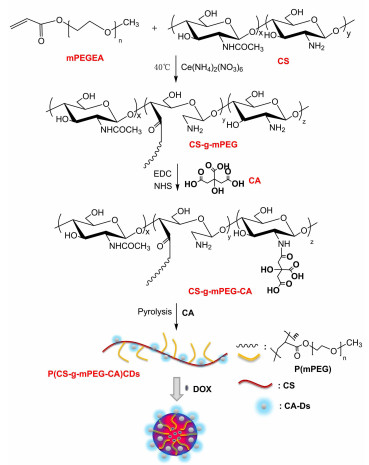
摘要: 荧光碳点具有化学稳定性好、毒性小、可表面功能化等优点,引起了人们极大的兴趣。近年来,由高分子多糖合成的聚合物碳点成为另一研究热点。本文通过水热法合成了一种壳聚糖基荧光聚合物碳点材料(P(CS-g-mPEG-CA)CDs),并用于载药研究。基于壳聚糖和聚乙二醇既是碳点的碳源也是碳点的钝化试剂,本文选择壳聚糖接枝聚乙二醇单甲醚和柠檬酸衍生物作为聚合物碳点的碳源,以提高聚合碳点的量子产率。另外,聚合物碳点还可以保留聚乙二醇与壳聚糖分子结构,为其在载药方面的应用提供有利条件。采用红外光谱、紫外光谱、X射线衍射、光电子能谱、透射电子显微镜和光致发光光谱对P(CS-g-mPEG-CA)CDs进行了结构表征以及pH值稳定性的测试。结果表明,所合成的P(CS-g-mPEG-CA)CDs具有较高的荧光量子产率(66.81%)、较长的荧光寿命(15.247 ns)、良好的pH稳定性。以阿霉素为模型药物,利用该聚合物碳点进行了负载研究,结果表明,当聚乙二醇单甲醚取代度为11.9%时,聚合物碳点的载药量最高为51.3%,最大药物释放率为28.7%,此外,药物的装载和释放可以通过mPEG的接枝率进行控制。采用MTT法评价了聚合物的碳点对鼻咽癌细胞(CNE-2)的毒性作用。研究表明,空白聚合物碳点无明显细胞毒性,CNE-2细胞存活率随着载药胶束的增加而降低,说明载药胶束对CNE-2细胞有较强的抑制作用。可见该P(CS-g-mPEG-CA)CDs在荧光标记、药物递送、荧光示踪系统和控制释放方面,具有一定的应用前景。Abstract: Fluorescent carbon dots have the advantages of good chemical stability, low toxicity, and surface functionalization, which has caused concern. In recent years, polymer carbon dots synthesize by polymer polysaccharides have become a new research hotspot. In this paper, a chitosan-based fluorescent polymer carbon dot material is synthesized by hydrothermal method and used for drug-loading research. We choose chitosan-graft-polyethylene glycol monomethyl ether and citric acid derivatives as the carbon sources for the carbon dots, because chitosan and polyethylene glycol are both a carbon source for carbon dots and a passivation reagent for carbon dots. Then the quantum yield of the polymeric carbon dots is increased. Polymer carbon dots can also retain the molecular structure of polyethylene glycol and chitosan, providing favorable conditions for its application in drug loading. The structural characterization is performed on P(CS-g-mPEG-CA)CDs by IR, UV, X-ray diffraction, photoelectron spectroscopy, transmission electron microscopy and photoluminescence spectra and pH stability test is carried out. The results show that the synthesized P(CS-g-mPEG-CA)CDs has higher fluorescence quantum yield(66.81%), longer fluorescence lifetime(15.247 ns), and better pH stability. Using Doxorubicin as a model drug, a load study was conducted using this polymer carbon dot. The results show that if the degree of substitution of mPEG is 11.9%, the maximum loading rate of polymer carbon dots is 51.3% and the maximum drug release rate is 28.7%. In addition, we also found that drug loading and release could be controlled by the grafting rate of mPEG. In addition, the cytotoxicity of polymer carbon dots on nasopharyngeal carcinoma cells(CNE-2) is evaluated using an MTT assay. The study shows that there is no obvious cytotoxicity of blank P(CS-g-mPEG-CA)CDs, and that the survival rate of CNE-2 cells decreases with the increase of drug-loaded micelles. The results show that the P(CS-g-mPEG-CA)CDs have a certain application prospect in the aspects of fluorescence labeling, drug delivery, fluorescent tracer system and controlled release.
-
Key words:
- chitosan /
- polymer carbon dots /
- citric acid /
- fluorescent materials /
- drug loading /
- mPEG /
- doxorubicin
-
Figure 5. UV-Vis spectrum and the maximum PL excitation and emission spectra of P CS-g-mPEG-CA) CDs in water and their digital photographs(a) under daylight and UV light(b)(A); PL emission spectra of P(CS-g-mPEG-CA)CDs under different wavelength excitations(B); Fluorescence lifetime(C) and fluorescence quantum yield(QY)(D) of the P(CS-g-mPEG-CA)CDs I, II, and III; Effect of the pH on the P(CS-g-mPEG-CA) CDs fluorescence, (all of the experiments were excited at 360nm)(E)
Table 1. Elemental analysis data, critical micelle concentration, DL and EE for different micelles
Samples C/N DSa of mPEG/% DL/% EE/% CMC/(μg·mL-1) Ⅰ 7.99 4.4 47.9 35.7 0.831 8 Ⅱ 10.52 8.2 49.4 39.6 5.623 4 Ⅲ 12.96 11.9 51.3 40.8 8.317 6 aDS:degree of substitution. -
[1] SUN Y P, ZHOU B, LIN Y, et al.. Quantum-sized carbon dots for bright and colorful photoluminescence[J]. Journal of the American Chemical Society, 2006, 128(24):7756-7762. doi: 10.1021/ja062677d [2] BAKER S N, BAKER G A. Luminescent carbon nanodots:emergent nanolights[J]. Angewandte Chemie International Edition, 2010, 49:6726-6744. doi: 10.1002/anie.200906623 [3] NⅡNO S, TAKESHITA S, ISO Y, et al.. Influence of chemical states of doped nitrogen on photoluminescence intensity of hydrothermally synthesized carbon dots[J]. Journal of Luminescence, 2016, 180:123-131. doi: 10.1016/j.jlumin.2016.08.021 [4] 娄庆, 曲松楠.基于超级碳点的水致荧光"纳米炸弹"[J].中国光学, 2015, 8(1):91-98. http://www.chineseoptics.net.cn/CN/abstract/abstract9260.shtmlLOU Q, QU S N. Water triggered luminescent'nano-bombs'based on supra-carbon-nanodots[J]. Chinese Optics, 2015, 8(1):91-98.(in Chinese) http://www.chineseoptics.net.cn/CN/abstract/abstract9260.shtml [5] HAN S, ZHANG H, XIE Y J, et al.. Application of cow milk-derived carbon dots/Ag NPs composite as the antibacterial agentn[J]. Applied Surface Science, 2015, 328:368-373. doi: 10.1016/j.apsusc.2014.12.074 [6] WANG C X, XU Z Z, CHENG H, et al.. A hydrothermal route to water-stable luminescent carbon dots as nanosensors for pH and temperature[J]. Carbon, 2015, 82:87-95. doi: 10.1016/j.carbon.2014.10.035 [7] BARUAH U, GOGOI N, MAJUMDAR G, et al.. β-Cyclodextrin and calix[4] arene-25, 26, 27, 28-tetrol capped carbon dots for selective and sensitive detection of fluoride[J]. Carbohydrate Polymers, 2015, 117:377-383. doi: 10.1016/j.carbpol.2014.09.083 [8] WANG W, CHENG L, LIU W. Biological applications of carbon dots[J]. Science China Chemistry, 2014, 57:522-539. doi: 10.1007/s11426-014-5064-4 [9] YAO J, YANG M, DUAN Y. Chemistry, biology, and medicine of fluorescent nanomaterials and related systems:new insights into biosensing, bioimaging, genomics, diagnostics, and therapy[J]. Chemical Reviews, 2014, 114:6130-6148. doi: 10.1021/cr200359p [10] XU X W, ZHANG K, ZHAO L, et al.. Aspirin-based Carbon dots, a good biocompatibility of material applied for bioimaging and anti-inflammation[J]. ACS Appl. Mater. Interfaces, 2016, 8:32706-32716. doi: 10.1021/acsami.6b12252 [11] TAO S Y, SONG Y B, ZHU S J, et al.. A new type of polymer carbon dots with high quantum yield:from synthesis to investigation on fluorescence mechanism[J]. Polymer, 2017, 116:472-478. doi: 10.1016/j.polymer.2017.02.039 [12] SONG G, LIN Y N, WANG H L. Strong fluorescence of poly(N-vinylpyrrolidone) and its oxidized hydrolysate[J]. Macromolecular Rapid Communications, 2015, 36:278-285. doi: 10.1002/marc.201400516 [13] ZHU S J, SONG Y B, SHAO J R, et al.. Non-conjugated polymer dots with crosslink-enhanced emission in the absence of fluorophore units[J]. Angewandte Chemie International Edition, 2015, 47:14626-14637. http://cn.bing.com/academic/profile?id=8e6e271cc5fb19bc846db8dc139ff7c9&encoded=0&v=paper_preview&mkt=zh-cn [14] YANG Y H, CUI J H, ZHENG M T, et al.. One-step synthesis of amino-functionalized fluorescent carbon nanoparticles by hydrothermal carbonization of chitosan[J]. Chemical Communications, 2012, 48:380-382. doi: 10.1039/C1CC15678K [15] XIAO D L, YUAN D H, HE H, et al.. Microwave-assisted one-step green synthesis of amino-functionalized fluorescent carbon nitride dots from chitosan[J]. Luminescence, 2013, 28:612-615. doi: 10.1002/bio.v28.4 [16] WANG, Y F, WANG X, GENG Z H, et al.. Electrodeposition of a carbon dots/chitosan composite produced by a simple in situ method and electrically controlled release of carbon dots[J]. Journal of Materials Chemistry B, 2015, 3:7511-7517. doi: 10.1039/C5TB01051A [17] ZU Y X, BI J R, YAN H P, et al.. Nanostructures derived from starch and chitosan for fluorescence bio-imaging[J]. Nanomaterials, 2016, 6:130-143. doi: 10.3390/nano6070130 [18] TANG Z J, LI G K, HU Y L. Advances in preparation and applications in quantitative analysis of nitrogen-doped carbon dots[J]. Progress in Chemistry, 2016, 28:1455-1461. http://cn.bing.com/academic/profile?id=e9000fef9c5aefc6345ee55badfaca52&encoded=0&v=paper_preview&mkt=zh-cn [19] LIN Y, ZHANG L Z, YAO W, et al.. Water-soluble chitosan-quantum dot hybrid nanospheres toward bioimaging and biolabeling[J]. ACS Applied Materials & Interfaces, 2011, 3:995-1002. http://cn.bing.com/academic/profile?id=693f8e0a4ecc3998f81c21e481a6590d&encoded=0&v=paper_preview&mkt=zh-cn [20] WADAJKAR A S, KADAPURE T, ZHANG Y, et al.. Dual-imaging enabled cancer-targeting nanoparticles[J]. Advanced Healthcare Materials, 2012, 1:450-456. doi: 10.1002/adhm.201100055 [21] CHEN G, WANG L W, CORDIE T, et al.. Multi-functional self-fluorescent unimolecular micelles for tumor-targeted drug delivery and bioimaging[J]. Biomaterials, 2015, 47:41-50. doi: 10.1016/j.biomaterials.2015.01.006 [22] WU D Q, LU B, CHANG C, et al.. Galactosylated fluorescent labeled micelles as a liver targeting drug carrier[J]. Biomaterials, 2009, 30:1363-1371. doi: 10.1016/j.biomaterials.2008.11.027 [23] LEE Y K, HONG S M, KIM J S, et al.. Encapsulation of CdSe/ZnS quantum dots in poly(ethylene glycol)-Poly(D, L-lactide) micelle for biomedical imaging and detection[J]. Macromolecular Research, 2007, 15:330-336. doi: 10.1007/BF03218795 [24] SILL K, EMRICK T. Nitroxide-mediated radical polymerization from CdSe nanoparticles micelles[J]. Chemistry of Materials, 2004, 16:1240-1243. doi: 10.1021/cm035077b [25] CHOWDHURI A R, TRIPATHY S, HALDAR C, et al.. Single step synthesis of carbon dot embedded chitosan nanoparticles for cell imaging and hydrophobic drug delivery[J]. Journal of Materials Chemistry B, 2015, 3:9122-9131. doi: 10.1039/C5TB01831E [26] RADHAKUMARY C, NAIR P D, NAIR C P R, et al.. Chitosan-comb-graft-polyethylene glycol monomethacrylate-synthesis, characterization, and evaluation as a biomaterial for hemodialysis applications[J]. Journal of Applied Polymer Science, 2009, 114:2873-2886. doi: 10.1002/app.v114:5 [27] KONO H, TESHIROGI T. Cyclodextrin-grafted chitosan hydrogels for controlled drug delivery[J]. International Journal of Biological Macromolecules, 2015, 72:299-308. doi: 10.1016/j.ijbiomac.2014.08.030 [28] GEDDA G, LEE C Y, LIN Y C, et al.. Green synthesis of carbon dots from prawn shells for highly selective and sensitive detection of copper ions[J]. Sensors and Actuators B:Chemical, 2016, 224:396-403. doi: 10.1016/j.snb.2015.09.065 [29] FU D J, JIN Y, XIE M Q, et al.. Preparation and characterization of mPEG grafted chitosan micelles as 5-fluorouracil carriers for effective anti-tumor activity[J]. Chinese Chemical Letters, 2014, 25:1435-1440. doi: 10.1016/j.cclet.2014.06.027 [30] PAPADIMITRIOU S A, ACHILIAS D S, BIKIARIS D N. Chitosan-g-PEG nanoparticles ionically crosslinked with poly(glutamic acid) and tripolyphosphate as protein delivery systems[J]. International Journal of Pharmaceutics, 2012, 430:318-327. doi: 10.1016/j.ijpharm.2012.04.004 [31] LI X Y, KONG X Y, SHI S, et al.. Biodegradable MPEG-g-chitosan and methoxy poly(ethylene glycol)-b-poly(e-caprolactone) composite films:Part 1.preparation and characterization[J]. Carbohydrate Polymers, 2010, 79:429-436. doi: 10.1016/j.carbpol.2009.08.032 [32] ZHANG Y, WANG Y L, FENG X T, et al.. Effect of reaction temperature on structure and fluorescence properties of nitrogen-doped carbon dots[J]. Applied Surface Science, 2016, 387:1236-1246. doi: 10.1016/j.apsusc.2016.07.048 [33] YANG S W, SUN, J, LI X B, et al.. Large-scale fabrication of heavy doped carbon quantum dots with tunable-photoluminescence and sensitive fluorescence detection[J]. Journal of Materials Chemistry A, 2014, 2:8660-8667. doi: 10.1039/c4ta00860j [34] ARDEKANI S M, DEHGHANI A, HASSAN M, et al.. Two-photon excitation triggers combined chemo-photothermal therapy via doped carbon nanohybrid dots for effective breast cancer treatment[J]. Chemical Engineering Journal, 2017, 330:651-662. doi: 10.1016/j.cej.2017.07.165 [35] FAN R J, SUN Q, ZHANG L, et al.. Photoluminescent carbon dots directly derived from polyethylene glycol and their application for cellular imaging[J]. Carbon, 2014, 71:87-93. doi: 10.1016/j.carbon.2014.01.016 [36] REMANT K B C, THAPA B, XU P S. pH and redox dual responsive nanoparticle for nuclear targeted drug delivery[J]. Molecular Pharmaceutics, 2012, 9:2719-2729. doi: 10.1021/mp300274g -






 下载:
下载: